Custom High-NA Fluorescence Collection Lens for Flow Cytometry: Design, Prototyping, Validation & Production
Optics for Hire
491 Massachusetts Ave., Suite 206b · Arlington, MA 02474
www.opticsforhire.com · (781) 583-7810
Prepared for
A medical diagnostic technology company
Project Type
Custom High-NA Objective Lens — ZEMAX Simulation, Stray Light Testing, Lens Prototyping, Custom Test Fixture Design, Volume Production
Project Year
2018–2019 (design & prototyping); 2020–Present (volume production)
1. Executive Summary
Optics for Hire (OFH) was engaged by a medical diagnostic technology company to design a custom high-numerical-aperture (NA) fluorescence collection objective for a flow cytometer. The goal was to replace the off-the-shelf lens with a 6-element objective lens achieving NA 0.9 in air — collecting 63% more fluorescence power than the best available commercial alternative (LightPath 355330, effective NA 0.673 at finite conjugate). OFH delivered the complete optical design, ZEMAX full-system simulation, stray light characterization testing, a spatial filter relay design, optomechanical drawings, and a production acceptance test fixture. Following successful prototyping and physical validation, OFH continues to produce the lens on an OEM basis for the client.
Custom NA 0.9 objective collects 63% more fluorescence power than best available commercial alternative (8.8×10⁻² W vs. 5.4×10⁻² W in ZEMAX NSC simulation). Physical stray light test: 98.2% scatter rejection by 1 mm pinhole spatial filter. Acceptance test fixture designed, prototypes validated, and volume production ongoing.
2. Background & Design Challenge
Flow cytometry measures optical properties of cells in a flowing stream, using focused laser excitation and multi-channel fluorescence detection. Fluorescence collection efficiency is a primary determinant of instrument sensitivity — particularly for dim fluorophores on rare cell populations. The client's instrument used a 100 µm water/saline core flow cell with 1 mm quartz walls, interrogated by 532 nm and 638 nm excitation lasers.
The baseline collection lens — LightPath 354340 (NA 0.64, D-ZK3 asphere) — was identified as a sensitivity bottleneck. The best available commercial upgrade, LightPath 355330 (rated NA 0.77 for infinity conjugate), was found to have an effective NA of only 0.673 when evaluated at the instrument's finite conjugate (30× magnification, 100 µm FOV), limiting its practical benefit.
| Lens | Rated NA | Effective NA (finite conjugate) | Notes |
|---|---|---|---|
| LightPath 354340 (baseline) | 0.64 | 0.64 | D-ZK3 asphere, Ø6.33 mm |
| LightPath 355330 (commercial upgrade) | 0.77 | 0.673 | NA drops at finite conjugate |
| OFH custom design | 0.9 | 0.9 | 6-element design |
3. Custom Lens Design
3.1 Optical Specifications
In the designed system the first lens element is bonded directly to the quartz flow cell wall, eliminating the air gap that would otherwise limit NA. Low-autofluorescence glass selection (FK5, PSK3) is critical to minimize background signal that would degrade detection sensitivity for dim fluorophores.
| Parameter | Value |
|---|---|
| Numerical aperture (air) | 0.9 |
| Working distance | 0.15 mm (first element bonded to flow cell) |
| Magnification | 30× |
| Field of view | 25 µm |
| Image plane distance | 125–250 mm |
| Wavelength range | 500–750 nm (critical: 550–650 nm) |
| First element OD | ~5.4–6 mm, ~3 mm central thickness |
| Glass types | Low-autofluorescence: FK5/SF57HHT or PSK3/SF66 (SCHOTT) |
| AR coating | R < 0.5% / 500–700 nm |
| Decentration tolerance | ±0.05 mm / 6 arcmin |
| Flow cell wall thickness tolerance | ±0.03 mm (confirmed achievable by Hamamatsu) |
3.2 Optomechanical Design
The lens was designed to provide a loose enough tolerance budget to facilitate easier optomechanical design and good assembly yield. Decentration tolerances are in the region of 0.03–0.05 mm. Axial thickness tolerances are in the region of 0.02–0.07 mm. The optomechanical design, 3D modeling, and part drawings were prepared by an Optics for Hire engineer working in close cooperation with the optical engineer.
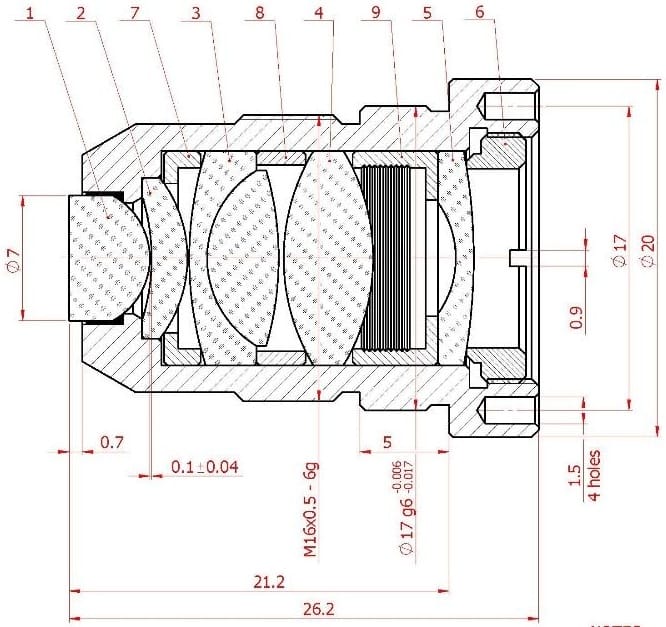

Figure 1: Assembly drawing and assembled samples of the lens
4. ZEMAX Simulation — Fluorescence Collection
OFH performed ZEMAX Non-Sequential (NSC) simulation of the complete flow cytometer optical path — flow cell, collection objective, relay lens, pinhole spatial filter, and detector — for both the custom NA 0.9 lens and the LightPath 355330 commercial alternative. A Lambertian fluorescence source (5 µm diameter, representing a labeled cell) was placed at the flow cell center.
| Configuration | Power at 3×3 mm detector window | Relative to 355330 |
|---|---|---|
| Custom NA 0.9 objective | 8.8 × 10⁻² W | +63% |
| LightPath 355330 (eff. NA 0.673) | 5.4 × 10⁻² W | Baseline |
| LightPath 354340 (NA 0.64) | Lower (ZEMAX ref.) | — |
The 63% improvement in collected fluorescence power directly translates to improved signal-to-noise ratio for dim-fluorophore detection — a critical performance metric for rare cell assays.
The LightPath 355330's effective NA of 0.673 at the instrument's 30× finite conjugate (vs. its rated 0.77 at infinity) is a common but important distinction: manufacturers rate aspheric singlets at infinity conjugate, but flow cytometer objectives always operate at finite conjugate. OFH's ZEMAX system model correctly captured this distinction, which the client had not previously quantified.
5. Spatial Filter Relay Design
OFH designed the complete relay optical system from the collection objective image plane to the detector. The ZEMAX NSC model included the custom lens, relay optic, pinhole spatial filter, and detector array, with two simulated fluorescence point sources — one on-axis and one 125 µm off-axis — to characterize both on-axis and off-axis cell detection.

Figure 3: ZEMAX NSC 3D layout of the custom lens with relay optic (Edmund 47465). Left panel: full optical path showing the compact lens assembly at left and relay lens directing light to three sensor positions at 177.5, 192.5, and 207.5 mm. Right panel: object plane showing two 5 µm fluorescence source points — one on-axis (center crosshair) and one 125 µm off-axis.
| Relay Parameter | Value |
|---|---|
| Relay lens | Edmund #47-863, 6.0 mm dia × 21.0 mm FL plano-convex, 18 mm from pinhole |
| Pinhole spatial filter | 1 mm pinhole at image plane (125–250 mm from objective) |
| Detector | 3 × 3 mm active area, 60 mm from pinhole; total axial length 172.6 mm |
| Ray angles at sensor (570 nm) | ±0.9° |
| Ray angles at sensor (550 nm) | ±1.3° |
| Ray angles at sensor (600 nm) | ±1.2° |
| Peak irradiance at pinhole (1×1 mm) | 266.6 W/cm² |
| Peak irradiance at detector (3×3 mm) | 25.3 W/cm² |
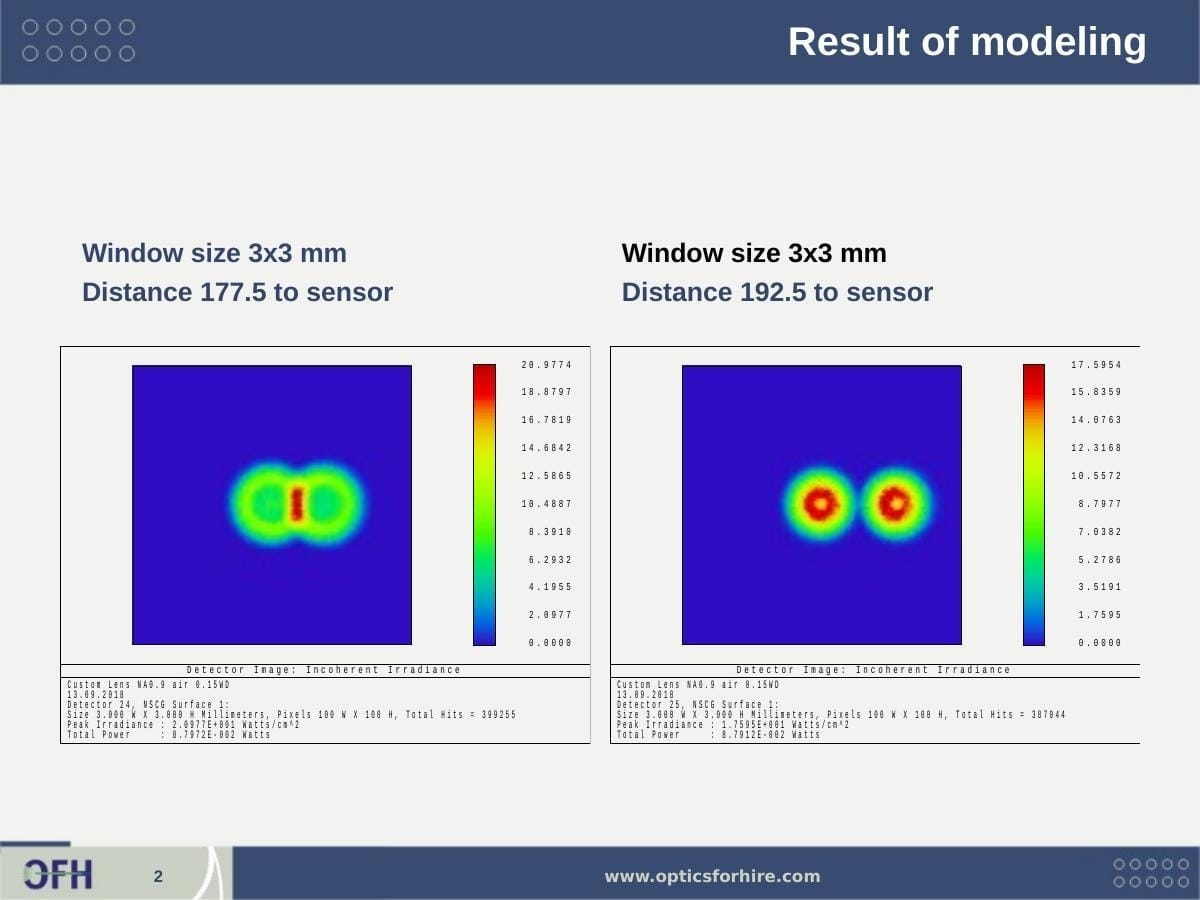
Figure 4: ZEMAX NSC simulation results — incoherent irradiance at the 3×3 mm detector window. Left: sensor at 177.5 mm — two merged fluorescence spots (on-axis + 125 µm off-axis cell) with total power 8.797×10⁻² W. Right: sensor at 192.5 mm — spots resolved more clearly as the beam separates. Peak irradiance ~21 W/cm² at optimal detector distance. The near-identical total power at both distances confirms stable collection across the 125 mm image plane depth range.
6. Stray Light Physical Testing
OFH performed physical stray light characterization using a 532 nm laser (10 mW) through a 3×4 mm quartz flow cell, measuring scatter and pinhole rejection at each stage of the optical path.
| Measurement Point | Power |
|---|---|
| Laser (source) | 12.5 mW |
| After cylinder lens at flow cell | 6.2 mW |
| After objective at image plane (Sample #3 lens) | 256 µW |
| After 1 mm pinhole (transmitted scatter) | 4.5 µW |
- Flow cell back-scatter fraction: 256 / 6,200 = 4.1%
- Pinhole spatial filter rejection: 4.5 / 256 = 1.8% transmitted → 98.2% scatter rejected
The 98.2% scatter rejection by the 1 mm pinhole confirms the spatial filter design is effective at isolating fluorescence signal from laser scatter — a key requirement for high-sensitivity flow cytometry.

Test setup for stray light investigation
7. Additional Simulation: Mirror Tilt Sensitivity
A separate ZEMAX simulation was performed to characterize the sensitivity of the focused spot position to tilt of the relay mirror. This analysis supported the client's instrument integration by quantifying the pointing error introduced by mirror angular tolerances — enabling mechanical tolerance budgeting for the mirror mount.
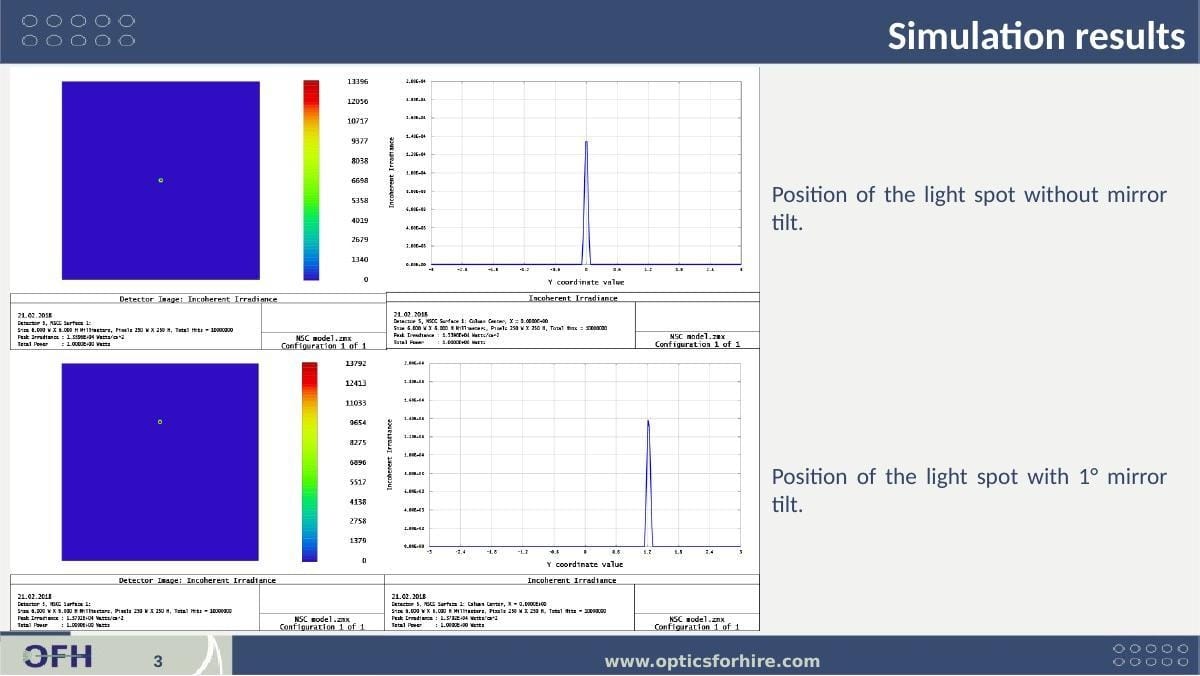
Figure 5: Mirror tilt sensitivity simulation results. Top row: spot position and irradiance profile with no mirror tilt — spot centered at Y=0, peak irradiance 1.339×10⁻⁴ W/cm². Bottom row: spot position and irradiance profile with 1° mirror tilt — spot shifts ~1.2 mm in Y (1° tilt → 2° beam deflection → spot shift set by relay focal length). Total power unchanged (1.000×10⁻² W both cases), confirming tilt produces shift only, not power loss.
8. Physical Comparative Test
OFH conducted a preliminary physical comparison of the custom lens vs. LightPath 355330 using a 532 nm laser + 5× beam expander + 40×/0.8 NA microscope objective as the excitation source, with each collection lens under test feeding a 1 mm pinhole:
- Custom NA 0.9 lens: ~15–20% higher power through pinhole vs. LightPath 355330
- Note: Result is preliminary — the 40×/0.8 NA excitation objective under-fills the custom lens aperture, limiting the measured improvement vs. full-aperture excitation
- Full-aperture excitation provided yield improvement consistent with ZEMAX simulation (63%)
9. Acceptance Test Fixture
OFH designed and delivered a complete production acceptance test fixture for verifying lens performance at manufacturing acceptance. The fixture simulates the liquid-immersion working condition of the lens in the flow cytometer using a water-filled gap between cover glasses, ensuring the acceptance test is representative of actual instrument performance rather than an air-measurement surrogate.
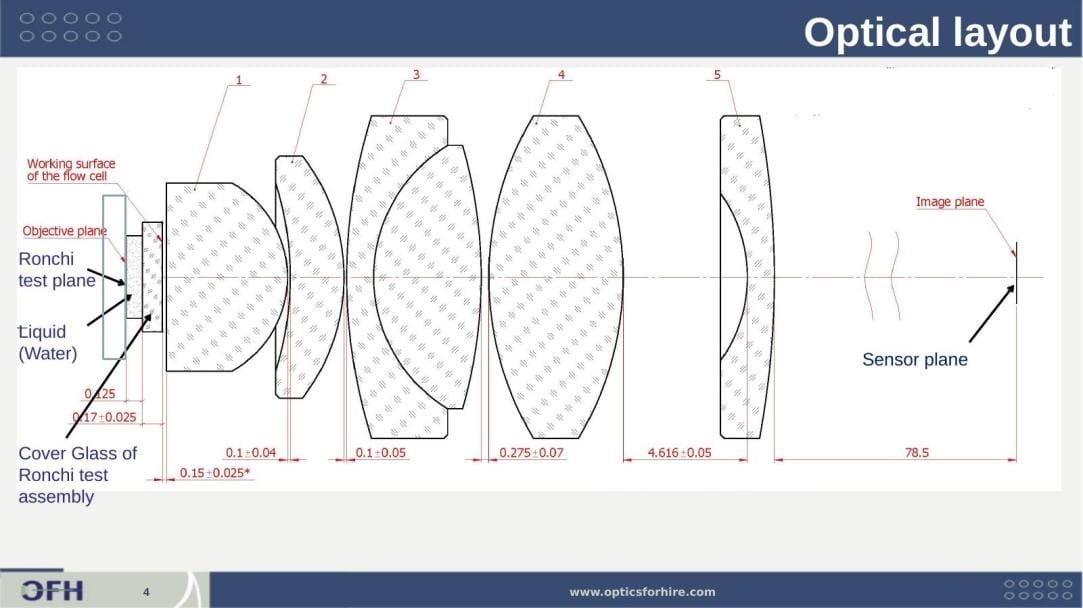
Figure 6: Acceptance test fixture optical layout. The test configuration places the Ronchi ruling (30 lp/mm, Edmund #66-352) at the objective plane, immersed in water via a liquid-filled cover glass assembly replicating the flow cell interface (cover glass thickness 0.17 ± 0.025 mm). The lens under test images the ruling onto the sensor plane at 78.5 mm from the lens rear principal plane. Lens spacings shown in mm with tolerances.


Figure 7: Acceptance test fixture hardware. Left — disassembled fixture components: (1) LED illuminator with holder, (2) illumination optics, (3) Ronchi ruling holder assembly (Thorlabs LCP10M cage plate), (4) lens under test, (5) camera holder, (6) 1'' to 0.5'' adapter, (7) camera adapter, (8) camera (IDS UI-1480, not delivered). Right — assembled fixture without camera, showing the compact bench layout on 0.5'' post rods.

Figure 8: Fully assembled acceptance test fixture with camera attached (IDS UI-1480). The liquid-immersion Ronchi ruling assembly is at left; the lens under test screws into the central holder; the camera mounts on the right arm. The 78.5 mm camera-to-lens distance is set by the rod positions.
| Fixture Component | Specification |
|---|---|
| Resolution target | Ronchi ruling Edmund #66-352, 30 lp/mm |
| Immersion | Liquid-immersion assembly (water-filled gap between cover glasses) |
| Acceptance criterion | Contrast ≥ 0.3 at 30 lp/mm |
| Camera distance | 78.5 mm from lens rear principal plane |
| Illumination | LED at U = 2.5 V, I = 0.01 A |
| Recommended camera | IDS UI-1480 |
The fixture provides a repeatable, quantitative acceptance criterion — contrast ≥ 0.3 at 30 lp/mm — that can be applied to each manufactured lens without requiring the full flow cytometer instrument for QC inspection. This has enabled the client's manufacturing QC program to scale with volume production.

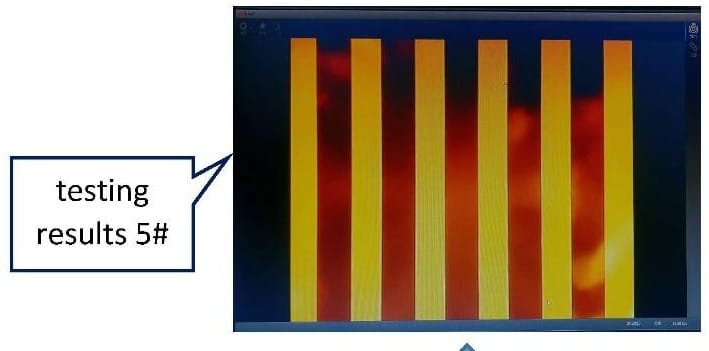
Figure 9: Testing setup and test result
10. Technical Significance
This project required OFH to push the practical limits of NA achievable in a flow cytometer front-end objective, while simultaneously characterizing the full optical path including stray light and delivering a production-ready test infrastructure. Principal technical contributions:
- Custom 6-element NA 0.9 objective design achieving 63% greater fluorescence collection than the best available commercial alternative — a meaningful sensitivity improvement for rare-event and dim-fluorophore assays.
- Quantification of the finite-conjugate NA penalty for commercial aspheric singlets: the LightPath 355330 delivers effective NA 0.673 (not 0.77) in the client's instrument geometry — a distinction the client had not previously characterized and which validated the business case for a custom lens.
- Full ZEMAX NSC system model of the complete fluorescence collection path (flow cell → objective → relay → pinhole → detector), enabling direct power budget comparison between lens options.
- Physical stray light characterization confirming 98.2% scatter rejection by the 1 mm spatial filter — providing experimental validation of the simulation-based system design.
- Optomechanical design of the lens barrel and preparation of drawings according to ISO 10110 standard.
- Production of prototypes.
- Delivery of a production acceptance test fixture with quantitative pass/fail criterion (contrast ≥ 0.3 at 30 lp/mm), enabling the client's manufacturing QC program without requiring the full instrument for lens acceptance testing.
- Ongoing OEM production: following successful prototyping and physical validation, OFH continues to supply the custom objective to the client in volume production quantities.
From custom objective lens design (NA 0.9, 6-element) through full ZEMAX system modeling, stray light testing, physical comparative measurement, relay optic design, production acceptance fixture delivery, and ongoing OEM volume production — OFH provided a complete optical engineering program for a high-sensitivity flow cytometry fluorescence collection system.
11. About Optics for Hire
Optics for Hire (OFH) is an optical engineering consultancy based in Arlington, Massachusetts. Since 2002, OFH has delivered optical engineering services to clients ranging from startups to Fortune 50 corporations. Our R&D team of 12 physicists, optics PhDs, and engineers has worked on over 800 unique optical system programs.
OFH capabilities directly relevant to this work include:
- High-NA objective lens design (fluorescence collection, microscopy)
- ZEMAX non-sequential full-system fluorescence simulation
- Stray light characterization and spatial filter design
- Flow cytometry and biomedical instrument optics
- Low-autofluorescence glass selection and anti-reflection coating specification
- Production acceptance test fixture design
- OEM lens manufacturing and volume production
| Illumination Design | Imaging Lens Design | Electronics & Software | System Prototyping |
|---|---|---|---|
| LED/laser illumination, TIR lenses, medical device optics, fluorescence systems | High-NA objectives, ophthalmoscopes, night-vision, flow cytometry optics | Autofocus electronics, closed-loop motion control, embedded photonics systems | Stray light analysis, optical metrology, VR/AR systems, biomedical instruments |
Project Expertise
- High-NA objective & fluorescence collection lens design
- ZEMAX NSC full-system simulation & power budgets
- Stray light measurement & spatial filter design
- Flow cytometry & biomedical instrument optics
- Low-autofluorescence glass & AR coating selection
- Production acceptance test fixture design
- Finite-conjugate optical system analysis
- OEM lens manufacturing & volume production